Detailed results of CTR107_XRay_em_bcr3 by PSVS
Output from PDBStat
Output from PROCHECK
Ramachandran Plot for all models
Text summary of Ramachandran Plot
+----------<<< P R O C H E C K S U M M A R Y >>>----------+
| |
| CTR107_XRay_em_bcr3_noHs_000.rin 0.0 153 residues |
| |
| Ramachandran plot: 95.9% core 4.1% allow 0.0% gener 0.0% disall |
| |
+| All Ramachandrans: 1 labelled residues (out of 151) |
+| Chi1-chi2 plots: 1 labelled residues (out of 77) |
JPEG image for all model Ramachandran Plot

Residue Properties for all models
JPEG for all model Residue Properties - page $num_n

JPEG for all model Residue Properties - page $num_n

JPEG for all model Residue Properties - page $num_n

Model Secondary Structures from Procheck
JPEG for Model Secondary Structures - page $num_n

JPEG for Model Secondary Structures - page $num_n

JPEG for Model Secondary Structures - page $num_n

Ramachandran Plots for each residue
JPEG for residue Ramachandran Plots - page $num_n

JPEG for residue Ramachandran Plots - page $num_n

JPEG for residue Ramachandran Plots - page $num_n

JPEG for residue Ramachandran Plots - page $num_n

JPEG for residue Ramachandran Plots - page $num_n

JPEG for residue Ramachandran Plots - page $num_n

JPEG for residue Ramachandran Plots - page $num_n

JPEG for residue Ramachandran Plots - page $num_n

Ramachandran analysis for each residue from Molprobity
Chi1-Chi2 Plots for each residue
JPEG for residue Chi1-Chi2 Plots - page $num_n

JPEG for residue Chi1-Chi2 Plots - page $num_n

JPEG for residue Chi1-Chi2 Plots - page $num_n

JPEG for residue Chi1-Chi2 Plots - page $num_n

Procheck G-factors for phi-psi for each residue
JPEG image for residue phi-psi G-factors
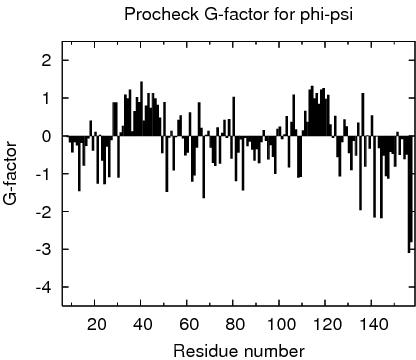
Table of Procheck G-factors for phi-psi for ordered residues
#phipsi_gfactor
#Residue\Model average
7 0.00
8 -0.03
9 -0.17
10 -0.43
11 -0.16
12 -0.25
13 -1.46
14 -0.18
15 -0.78
16 -0.26
17 -0.06
18 0.40
19 -0.39
20 0.10
21 -1.26
22 0.02
23 -0.65
24 -1.27
25 -0.28
26 -1.09
27 -0.11
28 0.88
29 0.88
30 -1.10
31 0.09
32 0.26
33 1.09
34 0.99
35 1.22
36 0.12
37 0.65
38 1.02
39 0.89
40 1.43
41 0.40
42 0.80
43 1.13
44 0.74
45 1.13
46 0.99
47 0.82
48 0.48
49 -0.45
50 0.89
51 -1.48
52 -0.04
53 0.13
54 -0.91
55 -0.03
56 0.42
57 0.54
58 -0.06
59 -0.51
60 -0.44
61 0.62
62 -1.21
63 -1.04
64 -0.31
65 0.88
66 0.21
67 -1.64
68 0.02
69 0.13
70 -0.31
71 -0.71
72 -0.79
73 0.22
74 -0.73
75 0.08
76 0.42
77 -0.05
78 0.44
79 -0.60
80 1.03
81 -1.19
82 -0.44
83 -0.09
84 -1.44
85 -0.05
86 -0.27
87 -0.15
88 -0.36
89 -0.65
90 -0.35
91 -0.72
92 -0.16
93 0.15
94 -0.13
95 -0.62
96 -0.24
97 -0.55
98 -1.00
99 0.18
100 0.24
101 -0.08
102 0.04
103 0.52
104 -0.83
105 0.37
106 1.09
107 0.17
108 -1.10
109 -1.08
110 0.14
111 0.66
112 0.37
113 1.22
114 1.32
115 0.99
116 1.13
117 0.84
118 1.22
119 1.26
120 0.98
121 1.08
122 0.30
123 -0.05
124 0.53
125 -0.56
126 -1.07
127 -0.16
128 0.43
129 0.25
130 -0.45
131 -0.90
132 -0.13
133 -0.52
134 0.35
135 -1.96
136 1.13
137 -0.81
138 0.01
139 -0.33
140 0.54
141 -2.15
142 -0.01
143 -0.32
144 -2.17
145 -0.52
146 -1.06
147 -1.13
148 -0.42
149 -0.47
150 -0.81
151 0.10
152 -0.50
153 -0.08
154 -0.61
155 -0.49
156 -3.09
157 -2.81
#Reported_Model_Average -0.107
#Overall_Average_Reported -0.107
Procheck G-factors for all dihedral angles for each residue
JPEG image for residue all dihedral G-factors
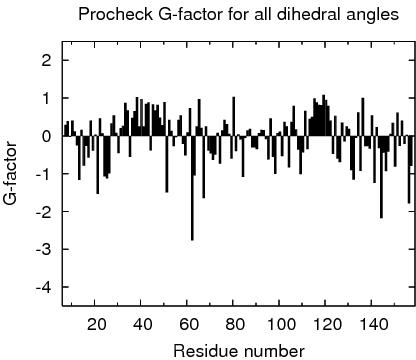
Table of Procheck G-factors for all dihedrals for ordered residues
#alldih_gfactor
#Residue\Model average
6 -2.70
7 0.29
8 0.39
9 -0.02
10 0.40
11 0.12
12 -0.25
13 -1.16
14 0.16
15 -0.78
16 -0.26
17 -0.57
18 0.40
19 -0.39
20 0.03
21 -1.53
22 0.46
23 0.06
24 -1.07
25 -1.12
26 -0.99
27 0.33
28 0.54
29 0.08
30 -0.45
31 0.20
32 0.26
33 0.87
34 0.67
35 -0.55
36 0.47
37 0.65
38 1.02
39 0.25
40 0.97
41 0.25
42 0.84
43 0.88
44 -0.38
45 0.83
46 0.67
47 0.82
48 0.48
49 0.28
50 0.89
51 -1.49
52 0.42
53 0.13
54 -0.27
55 -0.03
56 0.42
57 0.54
58 -0.21
59 -0.51
60 0.09
61 0.73
62 -2.76
63 -1.04
64 0.25
65 0.97
66 0.21
67 -1.64
68 0.25
69 -0.39
70 -0.46
71 -0.63
72 -0.49
73 0.08
74 -0.73
75 0.14
76 0.42
77 0.31
78 0.05
79 -0.60
80 1.03
81 -0.40
82 0.04
83 -0.09
84 -1.08
85 -0.05
86 0.14
87 0.18
88 -0.30
89 -0.30
90 -0.35
91 0.07
92 0.16
93 0.15
94 -0.08
95 -0.62
96 0.46
97 -0.55
98 -1.00
99 0.07
100 0.11
101 -0.53
102 0.37
103 0.25
104 -0.83
105 0.37
106 0.79
107 0.17
108 -0.36
109 -1.01
110 -0.43
111 0.66
112 -0.35
113 0.45
114 0.50
115 0.99
116 0.88
117 0.82
118 0.81
119 1.08
120 0.95
121 0.79
122 0.40
123 -0.47
124 0.53
125 -0.60
126 -0.69
127 0.35
128 -0.14
129 0.25
130 0.18
131 -0.90
132 -1.15
133 -0.05
134 0.62
135 -0.92
136 1.00
137 -0.27
138 -0.27
139 -0.33
140 0.54
141 -1.24
142 0.23
143 -0.32
144 -2.17
145 -0.44
146 -0.93
147 -0.41
148 0.05
149 0.34
150 -0.81
151 0.61
152 -0.26
153 0.40
154 -0.21
155 0.02
156 -1.78
157 -0.79
158 -0.72
#Reported_Model_Average -0.067
#Overall_Average_Reported -0.067
Output from Verify3D
Verify3D Score over a window of $winsize_s residues
JPEG image for Verify3D Score
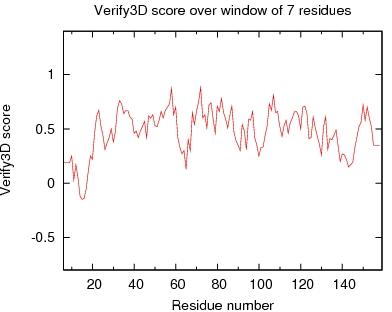
Table of Verify3D scores for ordered residues across all models
#verify3d
#Residue\Model only_model
6 -0.03
7 0.71
8 -0.80
9 1.28
10 0.41
11 -0.81
12 0.55
13 0.41
14 -0.81
15 0.14
16 0.44
17 -0.74
18 -1.01
19 0.59
20 1.07
21 0.36
22 1.07
23 0.19
24 0.95
25 0.10
26 0.95
27 0.08
28 -0.27
29 0.16
30 0.62
31 1.30
32 0.63
33 0.16
34 0.71
35 1.28
36 0.60
37 0.44
38 0.63
39 0.86
40 0.17
41 0.29
42 1.11
43 -0.30
44 0.62
45 0.16
46 1.30
47 0.44
48 0.63
49 0.10
50 1.10
51 0.47
52 0.34
53 0.64
54 0.34
55 1.10
56 0.64
57 0.64
58 0.96
59 0.59
60 0.84
61 1.30
62 -0.56
63 1.10
64 -1.30
65 0.34
66 0.14
67 1.10
68 0.08
69 1.32
70 0.41
71 1.18
72 -0.42
73 0.96
74 1.75
75 0.96
76 -0.65
77 0.66
78 0.28
79 1.10
80 1.10
81 0.66
82 0.04
83 1.10
84 0.34
85 1.10
86 0.24
87 0.66
88 0.08
89 0.79
90 1.75
91 -1.33
92 0.55
93 -0.07
94 0.34
95 1.75
96 0.35
97 0.59
98 0.59
99 0.49
100 0.49
101 -1.33
102 1.30
103 -0.35
104 1.10
105 0.64
106 1.25
107 1.10
108 0.04
109 0.93
110 0.60
111 -0.02
112 0.74
113 0.27
114 0.44
115 0.76
116 1.30
117 -0.27
118 0.66
119 1.01
120 0.74
121 0.44
122 0.44
123 0.51
124 1.10
125 0.71
126 0.51
127 -0.81
128 0.49
129 1.75
130 -0.20
131 0.59
132 0.17
133 -0.20
134 1.07
135 1.09
136 -0.33
137 0.51
138 0.51
139 0.44
140 0.14
141 0.04
142 0.08
143 0.14
144 0.44
145 0.23
146 -0.03
147 0.29
148 0.19
149 0.95
150 0.84
151 1.18
152 0.49
153 1.07
154 -0.68
155 1.07
156 0.20
157 0.28
158 0.00
#Reported_Model_Average 0.473
#Overall_Average_Reported 0.473
Output from ProsaII
ProsaII Score over a window of $winsize_s residues
JPEG image for ProsaII Score
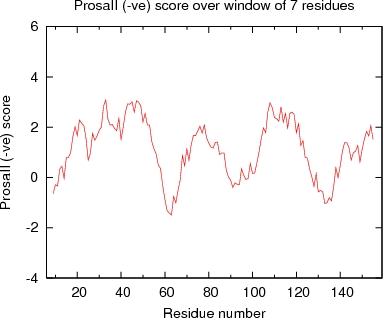
Table of Verify3D scores for ordered residues across all models
#verify3d
#Residue\Model only_model
6 -0.03
7 0.71
8 -0.80
9 1.28
10 0.41
11 -0.81
12 0.55
13 0.41
14 -0.81
15 0.14
16 0.44
17 -0.74
18 -1.01
19 0.59
20 1.07
21 0.36
22 1.07
23 0.19
24 0.95
25 0.10
26 0.95
27 0.08
28 -0.27
29 0.16
30 0.62
31 1.30
32 0.63
33 0.16
34 0.71
35 1.28
36 0.60
37 0.44
38 0.63
39 0.86
40 0.17
41 0.29
42 1.11
43 -0.30
44 0.62
45 0.16
46 1.30
47 0.44
48 0.63
49 0.10
50 1.10
51 0.47
52 0.34
53 0.64
54 0.34
55 1.10
56 0.64
57 0.64
58 0.96
59 0.59
60 0.84
61 1.30
62 -0.56
63 1.10
64 -1.30
65 0.34
66 0.14
67 1.10
68 0.08
69 1.32
70 0.41
71 1.18
72 -0.42
73 0.96
74 1.75
75 0.96
76 -0.65
77 0.66
78 0.28
79 1.10
80 1.10
81 0.66
82 0.04
83 1.10
84 0.34
85 1.10
86 0.24
87 0.66
88 0.08
89 0.79
90 1.75
91 -1.33
92 0.55
93 -0.07
94 0.34
95 1.75
96 0.35
97 0.59
98 0.59
99 0.49
100 0.49
101 -1.33
102 1.30
103 -0.35
104 1.10
105 0.64
106 1.25
107 1.10
108 0.04
109 0.93
110 0.60
111 -0.02
112 0.74
113 0.27
114 0.44
115 0.76
116 1.30
117 -0.27
118 0.66
119 1.01
120 0.74
121 0.44
122 0.44
123 0.51
124 1.10
125 0.71
126 0.51
127 -0.81
128 0.49
129 1.75
130 -0.20
131 0.59
132 0.17
133 -0.20
134 1.07
135 1.09
136 -0.33
137 0.51
138 0.51
139 0.44
140 0.14
141 0.04
142 0.08
143 0.14
144 0.44
145 0.23
146 -0.03
147 0.29
148 0.19
149 0.95
150 0.84
151 1.18
152 0.49
153 1.07
154 -0.68
155 1.07
156 0.20
157 0.28
158 0.00
#Reported_Model_Average 0.473
#Overall_Average_Reported 0.473
Output from MolProbity
VdW violations from MAGE
JPEG image for MAGE VdW violation
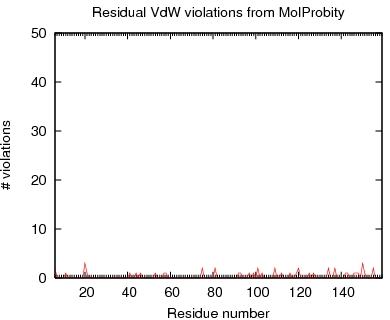
Table of MAGE VdW violations for ordered residues across all models
#mage_clash
#Residue\Model only_model
6.000 2
7.000 0
8.000 0
9.000 0
10.000 0
11.000 1
12.000 0
13.000 0
14.000 0
15.000 0
16.000 0
17.000 0
18.000 0
19.000 0
20.000 3
21.000 1
22.000 0
23.000 0
24.000 0
25.000 0
26.000 0
27.000 0
28.000 0
29.000 0
30.000 0
31.000 0
32.000 0
33.000 0
34.000 0
35.000 0
36.000 0
37.000 0
38.000 0
39.000 0
40.000 0
41.000 1
42.000 0
43.000 0
44.000 1
45.000 0
46.000 1
47.000 0
48.000 0
49.000 0
50.000 0
51.000 0
52.000 0
53.000 1
54.000 0
55.000 0
56.000 0
57.000 1
58.000 1
59.000 0
60.000 0
61.000 0
62.000 0
63.000 0
64.000 0
65.000 0
66.000 0
67.000 0
68.000 0
69.000 0
70.000 0
71.000 0
72.000 0
73.000 0
74.000 0
75.000 2
76.000 0
77.000 0
78.000 0
79.000 0
80.000 0
81.000 2
82.000 0
83.000 0
84.000 0
85.000 0
86.000 0
87.000 0
88.000 0
89.000 0
90.000 0
91.000 0
92.000 1
93.000 1
94.000 0
95.000 0
96.000 0
97.000 1
98.000 0
99.000 1
100.000 0
101.000 2
102.000 0
103.000 1
104.000 0
105.000 0
106.000 0
107.000 0
108.000 0
109.000 2
110.000 0
111.000 0
112.000 1
113.000 0
114.000 0
115.000 0
116.000 1
117.000 0
118.000 0
119.000 1
120.000 2
121.000 0
122.000 0
123.000 0
124.000 0
125.000 1
126.000 0
127.000 1
128.000 0
129.000 0
130.000 0
131.000 0
132.000 0
133.000 0
134.000 2
135.000 0
136.000 0
137.000 2
138.000 0
139.000 0
140.000 0
141.000 0
142.000 1
143.000 1
144.000 0
145.000 0
146.000 1
147.000 1
148.000 1
149.000 0
150.000 3
151.000 1
152.000 0
153.000 0
154.000 0
155.000 2
156.000 0
157.000 0
158.000 0
#Reported_Model_Average 0.314
#Overall_Average_Reported 0.314
List of bad contacts calculated by MAGE
/farm/software/bin/probe
: 2255:A 137 ASP 2HB :A 142 THR 1HG2 : -0.836: 35
: 2255:A 137 ASP 1HB :A 147 LEU 2HD1 : -0.705: 23
: 2255:A 134 ILE HB :A 150 ARG 2HB : -0.798: 17
: 2255:A 134 ILE 2HD1 :A 150 ARG 1HG : -0.446: 20
: 2255:A 99 SER OG :A 150 ARG 1HD : -0.432: 20
: 2255:A 58 PHE HZ :A 21 LEU 1HD2 : -0.710: 24
: 2255:A 41 ASP O :A 44 GLN 2HG : -0.614: 23
: 2255:A 109 ILE O :A 112 VAL 2HG1 : -0.604: 16
: 2255:A 151 VAL CG2 :A 109 ILE 1HD1 : -0.468: 19
: 2255:A 75 PHE 2HB :A 53 PRO 2HB : -0.557: 20
: 2255:A 75 PHE CE1 :A 57 PRO 2HB : -0.491: 17
: 2255:A 120 VAL 1HG2 :A 127 LEU 1HD2 : -0.535: 27
: 2255:A 116 LEU O :A 120 VAL 3HG2 : -0.434: 19
: 2255:A 97 ALA HA :A 155 LEU HG : -0.521: 19
: 2255:A 155 LEU 3HD1 :A 125 PHE CG : -0.434: 27
: 2255:A 143 ALA 3HB :A 146 GLN OE1 : -0.509: 37
: 2255:A 103 ILE 1HG1 :A 148 ARG 1HG : -0.494: 22
: 2255:A 93 PRO 1HD :A 92 THR HB : -0.452: 20
: 2255:A 20 LEU 3HD2 :A 20 LEU HA : -0.445: 16
: 2255:A 46 LEU 3HD2 :A 81 VAL 1HG1 : -0.427: 34
: 2255:A 81 VAL 1HG1 :A 20 LEU 1HD1 : -0.418: 34
: 2255:A 119 TRP HH2 :A 11 LEU 1HB : -0.437: 26
: 2255:A 101 LEU C :A 101 LEU 3HD2 : -0.427: 17
: 2255:A 6 GLN 2HE2 :A 6 GLN 1HB : -0.416: 44
#sum2 ::10.64 clashscore : 10.44 clashscore B<40
#summary::2255 atoms:2204 atoms B<40:255944 potential dots:16000.0 A^2:24 bumps:23 bumps B<40:722.8 score
Output from PDB validation software
Summary from PDB validation
May. 9, 23:54:49 2013
Greetings,
[ Text modified to reflect that this was run under PSVS - Aneerban Bhattacharya: Dec 2005 ]
The following checks were made on :
-----------------------------------------
DISTANCES AND ANGLES
We have checked your intra and intermolecular distances and angles with the
procedures currently in place at PDB:
==> The following solvent molecules are further away than 3.5 Angstroms from
macromolecule atoms which are available for hydrogen bonding in the
asymmetric unit.
none
The coordinates for water molecules which could be translated back into
the asymmetric unit are listed. If you do not indicate otherwise we will
replace the solvent coordinates in the entry with the ones below:
none
==> Close contacts in same asymmetric unit. Distances smaller than 2.2
Angstroms are considered as close contacts.
none
==> Close contacts based on crystal symmetry. Distances smaller than 2.2
Angstroms are considered as close contacts.
none
==> Bond and angle checks are performed by first computing the average rms
error for all bonds and angles relative to standard values for nucleotide
units [L. Clowney et al., Geometric Parameters in Nucleic Acids: Nitrogenous
Bases, J.Am.Chem.Soc. 1996, 118, 509-518; A. Gelbin et al., Geometric
Parameters in Nucleic Acids: Sugar and Phosphate Constituents, J.Am.Chem.Soc.
1996, 118, 519-529] and amino acid units [R.A. Engh and R. Huber, Accurate
Bond and Angle Parameters for X-ray protein structure refinement, Acta
Crystallogr. 1991, A47, 392-400]. Any bond or angle which deviates from the
dictionary values by more than six times this computed rms error is
identified as an outlier.
*** Covalent Bond Lengths:
The RMS deviation for covalent bonds relative to the standard
dictionary is 0.006 Angstroms
All covalent bonds lie within a 6.0*RMSD range about the
standard dictionary values.
*** Covalent Angle Values:
The RMS deviation for covalent angles relative to the standard
dictionary is 1.4 degrees.
The following table contains a list of the covalent bond angles
greater than 6.0*RMSD.
Deviation Residue Chain Sequence AT1 - AT2 - AT3 Bond Dictionary
Name ID Number Angle Value
--------------------------------------------------------------------------------
-11.1 LEU A 20 N - CA - C 100.1 111.2
-10.5 ILE A 22 N - CA - C 100.7 111.2
-9.8 PRO A 76 N - CA - C 102.0 111.8
-9.4 THR A 92 N - CA - C 101.8 111.2
-9.8 ALA A 98 N - CA - C 101.4 111.2
TORSION ANGLES
The torsion angle distributions have been checked. The postscript file of the
conformation rings showing the torsion angle distributions will be sent in a
separate E-mail message.
CHIRALITY
The chirality has been checked and there are no incorrect carbon chiral centers.
Some of O1P and O2P atoms do not follow the convention defined in the standard
IUBMB nomenclature (Liebecq, C. Compendium of Biochemical Nomenclature and Related
Documents, 2nd ed.; Portland Press: London and Chapel Hill, 1992). If you do not
indicate otherwise, we will switch the labels of O1P and O2P as shown below.
OTHER IMPORTANT ISSUES
==> Please check carefully REMARKS 3 and 200 and fill in the parameters as
appropriate.
==> The following residues are missing:
(Note: The SEQ number starts from 1 for each chain according to SEQRES
sequence record.)
RES MOD#C SEQ
MSE( A-152 )
ASP( A-151 )
PHE( A-150 )
GLU( A-149 )
CYS( A-148 )
GLN( A-147 )
PHE( A-146 )
VAL( A-145 )
CYS( A-144 )
GLU( A-143 )
LEU( A-142 )
LYS( A-141 )
GLU( A-140 )
LEU( A-139 )
ALA( A-138 )
PRO( A-137 )
VAL( A-136 )
PRO( A-135 )
ALA( A-134 )
LEU( A-133 )
LEU( A-132 )
ILE( A-131 )
ARG( A-130 )
THR( A-129 )
GLN( A-128 )
THR( A-127 )
THR( A-126 )
MSE( A-125 )
SER( A-124 )
GLU( A-123 )
LEU( A-122 )
GLY( A-121 )
SER( A-120 )
LEU( A-119 )
PHE( A-118 )
GLU( A-117 )
ALA( A-116 )
GLY( A-115 )
TYR( A-114 )
HIS( A-113 )
ASP( A-112 )
ILE( A-111 )
LEU( A-110 )
GLN( A-109 )
LEU( A-108 )
LEU( A-107 )
ALA( A-106 )
GLY( A-105 )
GLN( A-104 )
GLY( A-103 )
LYS( A-102 )
SER( A-101 )
PRO( A-100 )
SER( A -99 )
GLY( A -98 )
PRO( A -97 )
PRO( A -96 )
PHE( A -95 )
ALA( A -94 )
ARG( A -93 )
TYR( A -92 )
PHE( A -91 )
GLY( A -90 )
MSE( A -89 )
SER( A -88 )
ALA( A -87 )
GLY( A -86 )
THR( A -85 )
PHE( A -84 )
GLU( A -83 )
VAL( A -82 )
GLU( A -81 )
PHE( A -80 )
GLY( A -79 )
PHE( A -78 )
PRO( A -77 )
VAL( A -76 )
GLU( A -75 )
GLY( A -74 )
GLY( A -73 )
VAL( A -72 )
GLU( A -71 )
GLY( A -70 )
SER( A -69 )
GLY( A -68 )
ARG( A -67 )
VAL( A -66 )
VAL( A -65 )
THR( A -64 )
GLY( A -63 )
LEU( A -62 )
THR( A -61 )
PRO( A -60 )
SER( A -59 )
GLY( A -58 )
LYS( A -57 )
ALA( A -56 )
ALA( A -55 )
SER( A -54 )
SER( A -53 )
LEU( A -52 )
TYR( A -51 )
ILE( A -50 )
GLY( A -49 )
PRO( A -48 )
TYR( A -47 )
GLY( A -46 )
GLU( A -45 )
ILE( A -44 )
GLU( A -43 )
ALA( A -42 )
VAL( A -41 )
TYR( A -40 )
ASP( A -39 )
ALA( A -38 )
LEU( A -37 )
MSE( A -36 )
LYS( A -35 )
TRP( A -34 )
VAL( A -33 )
ASP( A -32 )
ASP( A -31 )
ASN( A -30 )
GLY( A -29 )
PHE( A -28 )
ASP( A -27 )
LEU( A -26 )
SER( A -25 )
GLY( A -24 )
GLU( A -23 )
ALA( A -22 )
TYR( A -21 )
GLU( A -20 )
ILE( A -19 )
TYR( A -18 )
LEU( A -17 )
ASP( A -16 )
ASN( A -15 )
PRO( A -14 )
ALA( A -13 )
GLU( A -12 )
THR( A -11 )
ALA( A -10 )
PRO( A -9 )
ASP( A -8 )
GLN( A -7 )
LEU( A -6 )
ARG( A -5 )
THR( A -4 )
ARG( A -3 )
VAL( A -2 )
SER( A -1 )
LEU( A 0 )
MSE( A 1 )
LEU( A 2 )
HIS( A 3 )
GLU( A 4 )
SER( A 5 )
PDB Chain_ID: A
1 15
SEQRES: MSE ASP PHE GLU CYS GLN PHE VAL CYS GLU LEU LYS GLU LEU ALA
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
16 30
SEQRES: PRO VAL PRO ALA LEU LEU ILE ARG THR GLN THR THR MSE SER GLU
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
31 45
SEQRES: LEU GLY SER LEU PHE GLU ALA GLY TYR HIS ASP ILE LEU GLN LEU
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
46 60
SEQRES: LEU ALA GLY GLN GLY LYS SER PRO SER GLY PRO PRO PHE ALA ARG
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
61 75
SEQRES: TYR PHE GLY MSE SER ALA GLY THR PHE GLU VAL GLU PHE GLY PHE
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
76 90
SEQRES: PRO VAL GLU GLY GLY VAL GLU GLY SER GLY ARG VAL VAL THR GLY
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
91 105
SEQRES: LEU THR PRO SER GLY LYS ALA ALA SER SER LEU TYR ILE GLY PRO
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
106 120
SEQRES: TYR GLY GLU ILE GLU ALA VAL TYR ASP ALA LEU MSE LYS TRP VAL
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
121 135
SEQRES: ASP ASP ASN GLY PHE ASP LEU SER GLY GLU ALA TYR GLU ILE TYR
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
136 150
SEQRES: LEU ASP ASN PRO ALA GLU THR ALA PRO ASP GLN LEU ARG THR ARG
COORDS: ... ... ... ... ... ... ... ... ... ... ... ... ... ... ...
151 165
SEQRES: VAL SER LEU MSE LEU HIS GLU SER GLN PHE VAL CYS GLU LEU LYS
COORDS: ... ... ... ... ... ... ... ... GLN PHE VAL CYS GLU LEU LYS
6 12
166 180
SEQRES: GLU LEU ALA PRO VAL PRO ALA LEU LEU ILE ARG THR GLN THR THR
COORDS: GLU LEU ALA PRO VAL PRO ALA LEU LEU ILE ARG THR GLN THR THR
13 27
181 195
SEQRES: MET SER GLU LEU GLY SER LEU PHE GLU ALA GLY TYR HIS ASP ILE
COORDS: MET SER GLU LEU GLY SER LEU PHE GLU ALA GLY TYR HIS ASP ILE
28 42
196 210
SEQRES: LEU GLN LEU LEU ALA GLY GLN GLY LYS SER PRO SER GLY PRO PRO
COORDS: LEU GLN LEU LEU ALA GLY GLN GLY LYS SER PRO SER GLY PRO PRO
43 57
211 225
SEQRES: PHE ALA ARG TYR PHE GLY MET SER ALA GLY THR PHE GLU VAL GLU
COORDS: PHE ALA ARG TYR PHE GLY MET SER ALA GLY THR PHE GLU VAL GLU
58 72
226 240
SEQRES: PHE GLY PHE PRO VAL GLU GLY GLY VAL GLU GLY SER GLY ARG VAL
COORDS: PHE GLY PHE PRO VAL GLU GLY GLY VAL GLU GLY SER GLY ARG VAL
73 87
241 255
SEQRES: VAL THR GLY LEU THR PRO SER GLY LYS ALA ALA SER SER LEU TYR
COORDS: VAL THR GLY LEU THR PRO SER GLY LYS ALA ALA SER SER LEU TYR
88 102
256 270
SEQRES: ILE GLY PRO TYR GLY GLU ILE GLU ALA VAL TYR ASP ALA LEU MET
COORDS: ILE GLY PRO TYR GLY GLU ILE GLU ALA VAL TYR ASP ALA LEU MET
103 117
271 285
SEQRES: LYS TRP VAL ASP ASP ASN GLY PHE ASP LEU SER GLY GLU ALA TYR
COORDS: LYS TRP VAL ASP ASP ASN GLY PHE ASP LEU SER GLY GLU ALA TYR
118 132
286 300
SEQRES: GLU ILE TYR LEU ASP ASN PRO ALA GLU THR ALA PRO ASP GLN LEU
COORDS: GLU ILE TYR LEU ASP ASN PRO ALA GLU THR ALA PRO ASP GLN LEU
133 147
301 311
SEQRES: ARG THR ARG VAL SER LEU MET LEU HIS GLU SER
COORDS: ARG THR ARG VAL SER LEU MET LEU HIS GLU SER
148 158
==> The following residues have missing atoms:
RES MOD#C SEQ ATOMS
MET( A 28) SD
MET( A 64) SD
MET( A 117) SD
MET( A 154) SD
==> The following residues have extra atoms:
RES MOD#C SEQ ATOMS
MET( A 28) SE
MET( A 64) SE
MET( A 117) SE
MET( A 154) SE
SER( A 158) O2
CTR107_XRay_em_bcr3.pdb: Error: Record (RES: MSE CHNID: A SSEQ: 28) in Token 'MODRES' can not be found in coordinates
CTR107_XRay_em_bcr3.pdb: Error: Record (RES: MSE CHNID: A SSEQ: 64) in Token 'MODRES' can not be found in coordinates
CTR107_XRay_em_bcr3.pdb: Error: Record (RES: MSE CHNID: A SSEQ: 117) in Token 'MODRES' can not be found in coordinates
CTR107_XRay_em_bcr3.pdb: Error: Record (RES: MSE CHNID: A SSEQ: 154) in Token 'MODRES' can not be found in coordinates
























